Ultragenyx Q2 Loss Shrinks, Revenues Jump Year-Over-Year
Ultragenyx Pharmaceutical Reports Improved Q2 2025 Performance
Ultragenyx Pharmaceutical, a biotechnology company focused on rare diseases, reported its second-quarter 2025 financial results, showing improved performance compared to expectations. The company recorded a loss of $1.17 per share, which is narrower than the Zacks Consensus Estimate of $1.27. This marks an improvement from the previous year’s loss of $1.52 per share.
Total revenues for the quarter reached $166.5 million, reflecting a 13% increase year over year. This outperformed the estimated revenue of $162 million. The growth was primarily driven by higher product sales, with all four of the company's marketed drugs contributing to the top line.
Key Products and Revenue Breakdown
Ultragenyx markets four drugs: Crysvita, Mepsevii, Dojolvi, and Evkeeza. Each has specific indications:
- Crysvita is approved for treating X-linked hypophosphatemia and tumor-induced osteomalacia.
- Mepsevii targets Mucopolysaccharidosis VII (Sly syndrome).
- Dojolvi is used for long-chain fatty acid oxidation disorders.
- Evkeeza is indicated for homozygous familial hypercholesterolemia (HoFH).
In 2022, Ultragenyx entered into a license and collaboration agreement with Regeneron Pharmaceuticals for Evkeeza. Under this deal, Ultragenyx obtained rights to develop, commercialize, and distribute the drug outside the U.S., while Regeneron retains commercialization rights in the U.S.
Detailed Q2 Performance
Crysvita generated $120.4 million in revenue during the second quarter, up 6% year over year. North America contributed $79 million, Latin America and Turkey added $35 million, and Europe brought in $7 million. Mepsevii saw a 35% increase in revenue to $8.3 million, while Dojolvi revenue rose 20% to $23.2 million. Evkeeza recorded $14.6 million in sales, reflecting strong performance as the company continues to expand its presence in international markets.
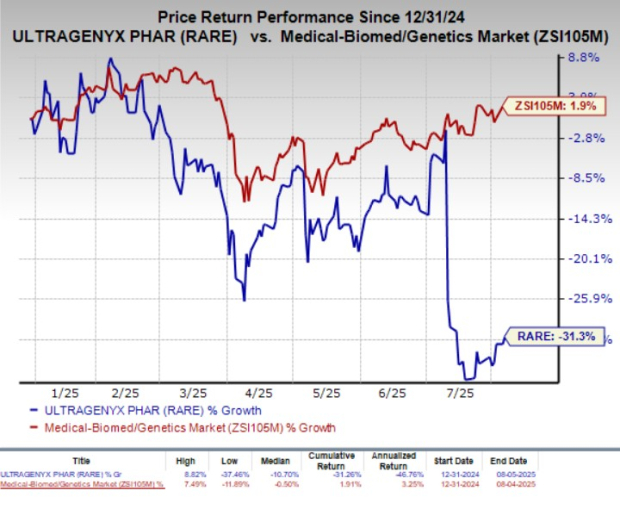
Year-to-date, shares of Ultragenyx have declined by 31.3%, significantly underperforming the industry’s 1.9% growth.
Operating Expenses and Financial Position
Operating expenses for the quarter were $274.4 million, a 4% increase from the previous year. This rise was attributed to increased investments in late-stage pipeline programs and marketing costs for approved drugs. Specifically, research and development expenses totaled $164.7 million (up 2%), selling, general, and administrative expenses reached $86.6 million (up 7%), and cost of sales increased to $23 million (up 8%).
As of June 30, 2025, the company held $539 million in cash, cash equivalents, and marketable debt securities, down slightly from $563 million at the end of March 2025.
2025 Financial Guidance
Ultragenyx reaffirmed its 2025 financial guidance, projecting total revenues between $640 million and $670 million, representing a 14-20% growth compared to 2024. Crysvita is expected to generate $460-$480 million in revenue, up 12-17% year over year, while Dojolvi is projected to bring in $90 million to $100 million, reflecting 2-14% growth.
The company plans to maintain focus on expense management while investing in upcoming commercial launches and advancing multiple phase III programs. These efforts are expected to reduce net cash used in operations in 2025 compared to 2024.
Pipeline Updates and Challenges
Last month, Ultragenyx faced a setback when the FDA issued a complete response letter (CRL) for its BLA for UX111, a treatment for MPS IIIA. The CRL requested additional information on chemistry, manufacturing, and controls, but the company believes the issues can be resolved quickly.
Ultragenyx and Mereo BioPharma are jointly developing UX143 (setrusumab) for osteogenesis imperfecta. The Orbit and Cosmic studies are on track for final analysis by the end of 2025.
Additionally, the FDA granted Breakthrough Therapy designation to GTX-102 for Angelman syndrome. A pivotal phase III study, Aspire, has completed enrollment, with data expected in the second half of 2026. An additional study, Aurora, is planned for 2025 to evaluate broader patient populations.
Ultragenyx also plans to submit a BLA for DTX401, an AAV8 gene therapy for glycogen storage disease type Ia, in the fourth quarter of 2025. The company is evaluating UX701, an AAV9 gene therapy, in a phase I/II/III trial for Wilson disease.
Zacks Rank and Market Outlook
Ultragenyx currently holds a Zacks Rank of #4 (Sell). In comparison, other biotech stocks like Arvinas (ARVN) and Immunocore (IMCR) carry a Zacks Rank of #1 (Strong Buy). ARVN has seen its 2025 loss estimates narrow to $1.50 from $1.51, while IMCR has narrowed its 2025 loss estimates to 68 cents from 86 cents. Both stocks have shown mixed performance, with ARVN declining sharply and IMCR rising modestly.

Posting Komentar untuk "Ultragenyx Q2 Loss Shrinks, Revenues Jump Year-Over-Year"
Posting Komentar